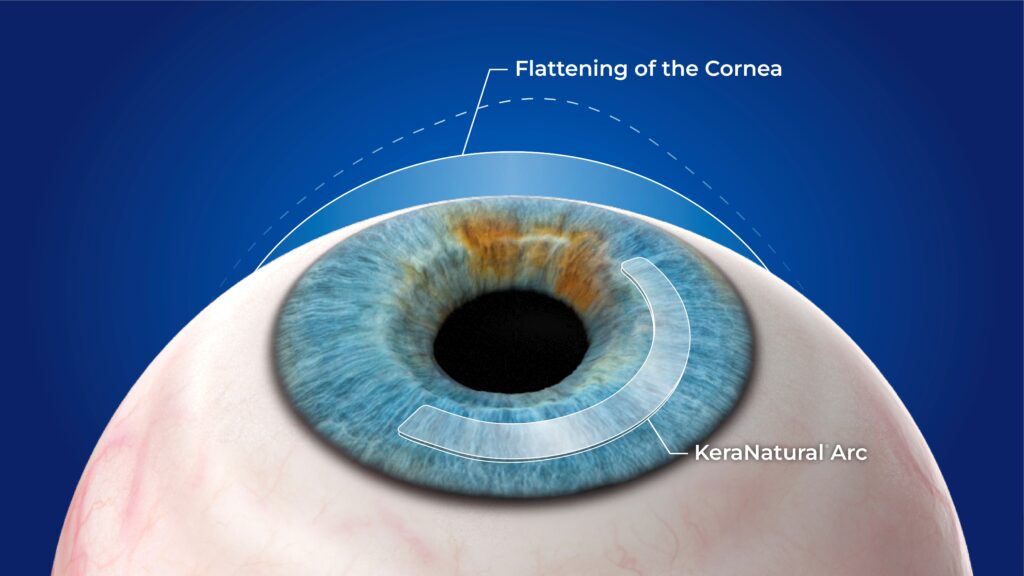
For surgeons who regularly manage keratoconus and corneal ectasia, INTACS and other intra-corneal ring segments have long played a role in stabilizing corneal shape and improving vision. As techniques continue to evolve, some surgeons are exploring CAIRS using KeraNatural as a next step in expanding treatment options for appropriate patients.
One surgeon recently shared insights from their experience of switching to KeraNatural after years of working with traditional ring segments.
A Natural Evolution
After using INTACS for many years, this surgeon viewed CAIRS with KeraNatural as a logical progression for their keratoconus and ectasia population.
“We were using INTACS for years, so it was a natural evolution to move into KeraNatural as a viable next step for our Keratoconus / ectasia population.”
Rather than replacing prior approaches, KeraNatural became an additional tool, particularly appealing for reducing certain ring-related complications.
The Learning Curve: Steep, but Manageable
The surgeon emphasized that implementing KeraNatural requires commitment and patience.
“There was a very steep learning curve in patient selection, surgical delivery, and outcomes analysis.”
Comfort with the procedure developed after approximately 15–20 cases, allowing the surgeon to better understand the nuances involved. This transparency is important for surgeons considering CAIRS, as it sets realistic expectations and underscores the importance of experience.
Training came through a combination of fellowship experience and support from the eye-bank team involved in tissue preparation and delivery.
Outcomes and Patient Experience
Once comfortable with the procedure, outcomes were described as tremendous, with a notable difference compared to previous approaches.
“The biggest difference between INTACS with CXL and KeraNatural is that patients are actually excited to have the second eye treated because they see the benefit and the ease of intervention.”
This shift in patient perception stood out. Whereas earlier treatments could feel arduous or underwhelming, patients treated with KeraNatural were more enthusiastic about continuing care.
Practical Tips and Best Practices
The surgeon also shared specific technical recommendations that proved beneficial in their workflow:
- Dehydrate the tissue for approximately 20 minutes in a clean environment.
- Use a Wek-Cel to assist with tissue deturgescence.
These small but important details helped optimize handling and outcomes.
Advice for Surgeons Considering KeraNatural
When asked what advice they would give colleagues exploring CAIRS, the response was straightforward:
“If you see, treat, and manage keratoconus and have an interest in providing a genuine tool for surgical improvement, then start treating your patients with KeraNatural.”
While reimbursement was initially a concern, experience and outcomes ultimately supported continued use.
Final Thoughts
This surgeon’s experience highlights several key takeaways:
- KeraNatural represents a meaningful evolution for surgeons familiar with intracorneal ring segments.
- A learning curve exists, but confidence develops with experience.
- Patient enthusiasm – particularly for second-eye treatment can be a powerful indicator of success.
- Practical technique refinements matter.
For surgeons managing keratoconus and ectasia, KeraNatural may offer a compelling option worth exploring as part of a comprehensive treatment strategy. As with any evolving technique, continued experience, peer discussion, and thoughtful implementation remain central to optimizing patient care.
The perspectives shared here are based on a surgeon’s firsthand experience and are published with permission for educational purposes. Surgeons interested in learning more or engaging in peer-to-peer discussions about KeraNatural may contact the Vision Share team for additional information.
About Vision Share
Vision Share is a 501(c)(3) nonprofit organization and proudly represents the world’s largest network of eye banks. Each member eye bank is Eye Bank Association of America (EBAA) certified and registered with the Food and Drug Administration (FDA). Guided by a shared vision, Vision Share pools resources to provide surgeons with dependable, high-quality ocular tissue tailored to their specific needs.
