Since endothelial keratoplasties were introduced to the United States in 2005, eye banks have relied on post-processing specular microscopy to estimate endothelial health—typically using a small central image that captures only a fraction of the graft and is often influenced by selection bias. Kerify, developed by Lions Gift of Sight (LGS) in partnership with imaging experts ADCIS and supported by the EBAA, represents a significant leap forward.
Kerify provides a fast, full-graft view with quantitative mapping of endothelial cell damage—bringing a level of precision and reproducibility that traditional methods cannot match. Using a small microscope stationed within the processing environment, Kerify also introduces speed and efficiency, representing an alluring innovation.
Recently, we spoke with the LGS team behind Kerify—Peter Bedard (Research Scientist), Amy Vallen (Director of Public and Partner Relations), and Brian Philippy (Director of Business Development)—to learn how the technology began, how it’s being used, and what it may mean for the future of eye banking and corneal transplantation.
How Kerify Began: From a Lab Tour to a Commercialized Platform.
Kerify’s story began six years ago when a local Lion, formerly a geology professor turned machine-vision engineer, toured the LGS lab in St. Paul, Minnesota and asked a simple question: Do you need help with machine vision?
At the time, many groups were focused on improving automated specular microscopy, but few were exploring ways to assess the entire cornea. ADCIS and the LGS team began designing and iterating software, using a variety of microscopes, for nearly a year, ultimately proving that full-graft imaging at the required resolution was feasible.
The newly developed optical system included ways to differentiate camera “noise,” and other visual distractions, like trypan back-staining, that computers could mistake for damage. Notably, Kerify didn’t begin with AI—artificial intelligence was incorporated later as imaging capabilities matured. The complete system, with both optical and AI-assisted analysis, was commercialized just last year.
The DEKS Study and the Push Toward Standardized Imaging
Kerify’s development has progressed alongside the Diabetes Endothelial Keratoplasty Study (DEKS) study — a multi-site, clinical study that aims to evaluate success of DMEK surgery based on donor and tissue characteristics, such as comprehesive endothelial damage. Because DEKS already had IRB approvals and active clinical sites, it provided an ideal framework for validating Kerify’s full-graft quantitative metrics against traditional damage estimates.
This alignment allowed the LGS team to launch a standardized imaging study comparing Kerify data with specular microscopy assessments and Weka segmentation, a pixel-level “gold standard.”
Study Goals Include:
- Capturing endothelial damage at multiple eye banks with a single protocol
- Comparing Kerify outputs with traditional specular-based estimates
- Determining how early damage correlates with graft failure and endothelial cell density loss at one to three years
The study has reached the one-year endpoint and the team will soon learn what the data reveals about the clinical impact of whole graft damage. The initial investigation will compare Kerify’s quantitative metrics with technician estimates and Weka segmentation analysis. A follow-up study by LGS in early 2026 will evaluate how Kerify metrics predict postoperative endothelial cell loss and graft failure.
How Does Kerify Fit Into Real-World Eye Bank Workflows?
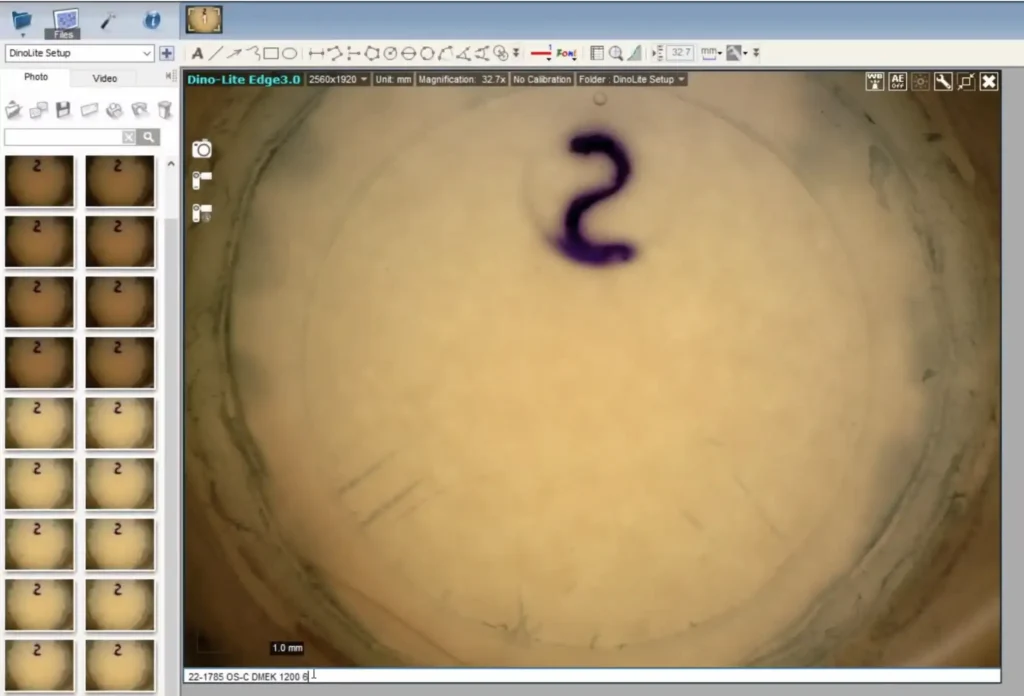
“It is our duty as stewards of these precious donated gifts to leverage technology to further evolve our capabilities in restoring sight. At LGS, we have been using Kerify to assess our DMEK-processed tissue for over a year now.
Since validating and implementing the system at our eye bank, we have appropriately prevented some grafts from use, identified proper utility of others, and ensured the best outcomes for our recipients. Surgeon response has been entirely positive.”
— Brian Philippy, Director of Business Development, LGS
During the launch phase, surgeons were notified about what to expect and how to interpret results. Surgeons responding to the communication applauded the innovation. Since implementation, the system has improved surgeon confidence in the evaluation process. Kerify adoption is growing. Three eye banks are currently using the system, and a fourth is onboarding.

One of the strongest drivers of adoption is workflow efficiency: unlike specular microscopy—which often requires tissue removal from a sterile field—Kerify allows imaging inside the hood.
This reduces handling time and saves an estimated 25–30 minutes per case. For eye banks that interrupt aseptic processing to remove tissue from the environment to evaluate, the change in procedure may also reduce or eliminate the need for a circulator.
What Are Surgeons Saying?
“There is a huge technology gap in how we transplant corneas and the antiquated method in which we select corneas for transplantation. Bringing our tissue evaluation technology up to par with modern techniques through Kerify is critical for improving patient outcomes.”— Joshua H. Hou, MD, Medical Director, LGS
Surgeons report increased trust in the evaluation process—particularly because Kerify eliminates a common frustration: cell density appearing higher after processing, which occurs about half the time with specular microscopy.
A More Complete Picture of Graft Quality
Specular microscopy has long-standing limitations:
- It assesses only a small central region
- Algorithms require ~100 contiguous cells
- To effectively operate the algorithm, technicians must select “cleaner” and healthier areas, introducing bias
Kerify overcomes these challenges by analyzing the entire graft and generating a quantitative damage map. This provides eye banks and surgeons with a more accurate, standardized, and clinically meaningful assessment.
As the DEKS ancillary work advances, the field will begin establishing widely adopted thresholds (e.g., what constitutes mild vs. moderate damage). In the meantime, Kerify’s team is normalizing ranges across many grafts and translating percentages into terminology that aligns with clinical decision-making.
Hardware, Software, and Adoption Ease
Kerify was designed to fit seamlessly into existing workflows:
- Hardware: A compact, high-resolution microscope that fits inside standard processing hoods.
- Software: Cloud-based and browser-accessible; users drag and drop images into a secure platform.
- Pricing: Available through a subscription or a pay-per-use models suitable for eye banks of all sizes.
Forms, Reporting, and Surgeon Communication
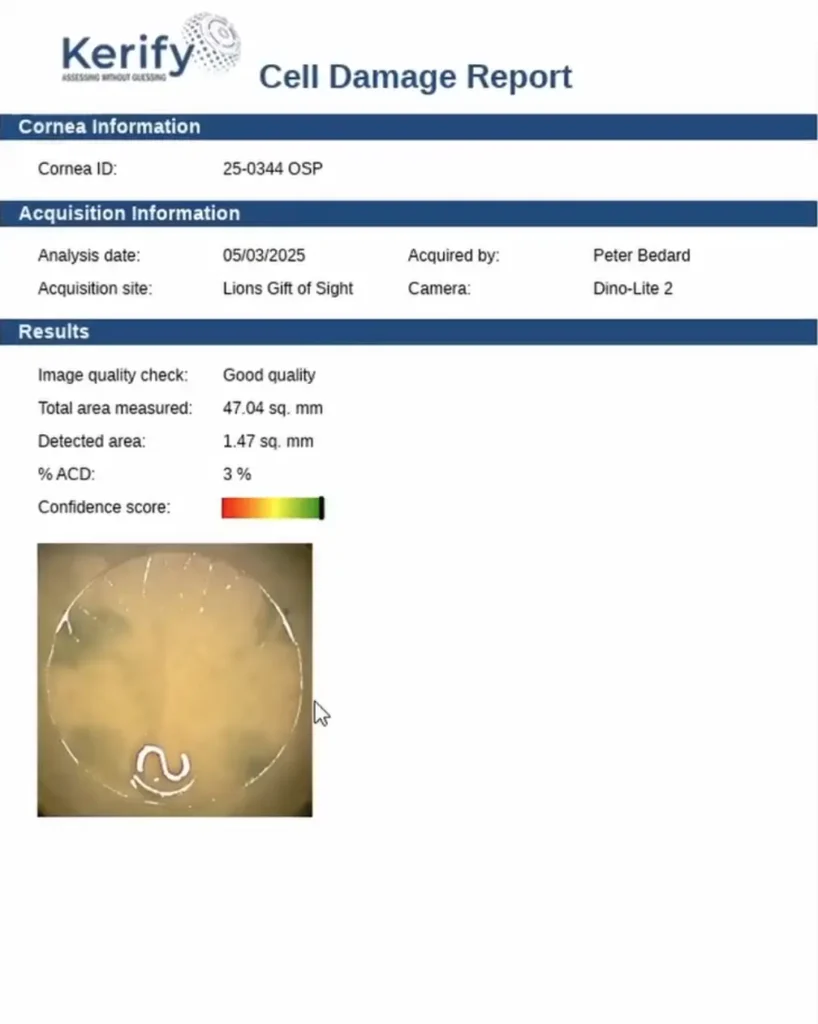
Eye banks accustomed to including specular images on processing forms can easily replace them with Kerify graft maps, including:
- Percentage of endothelial cell damage
- Interpretive category (e.g., mild, moderate)
The EBAA is evaluating emerging standards in this area. Initially, the team limited direct surgeon communication to avoid creating inconsistency across banks. However, surgeon education is becoming increasingly important as adoption grows. More surgeon-facing materials are in development.
What’s Next for Kerify?
Kerify’s next phase includes evaluating tissues in scroll or lenticular configurations—critical for DMEK and DSAEK— as well as imaging through injectors. These advancements will help eye banks generate workflow-specific data that better supports their surgical partners. For more information about Kerify or implementation, contact Brian Philippy, Director of Business Development at Lions Gift of Sight (email: phili013@umn.edu; phone 757-636-5563).
About Vision Share

Vision Share is a 501(c)(3) nonprofit organization and proudly represents the world’s largest network of eye banks. Each member eye bank is Eye Bank Association of America (EBAA) certified and registered with the Food and Drug Administration (FDA). Guided by a shared vision, Vision Share pools resources to provide surgeons with dependable, high-quality ocular tissue tailored to their specific needs. The Vision Share network has delivered over 141,000 corneas worldwide, transforming lives and restoring sight.
