As part of Vision Share’s ongoing series exploring breakthroughs in corneal transplantation, we recently met with Eversight’s Vice President of Research & Development, Onkar B. Sawant, PhD to discuss emerging innovations shaping the future of the field.
Together with insights from Eversight’s webinar featuring Dr. Edward Holland and Dr. Nambi Nallasamy, the conversation highlighted how science, technology, and eye-bank–driven solutions are redefining what is possible for patients worldwide.
This interview is part of Vision Share’s broader effort to collect diverse perspectives—from researchers, device developers, surgeons, and eye banks—on how innovation continues to transform corneal transplantation.
How Surgical Innovation Is Reshaping Corneal Care
Over the past decade, posterior lamellar techniques like DMEK have become the preferred surgical approach in appropriately selected cases for treating endothelial disease due to their faster recovery times, reduced rejection rates, and superior visual outcomes compared to traditional PKP.
For DMEK surgery, innovations such as the addition of an “S” stamp or other orientation markings to aid graft positioning, along with the preloading of DMEK tissue into injectors that are prepared by eye banks, represent important advancements that improve the efficiency of graft insertion into the anterior chamber. Together, these improvements have helped establish DMEK as a leading surgical option for standard endothelial failure in eyes with good anterior chamber visibility. ¹
These advances—paired with improved imaging, OR guidance technologies, and standardized graft preparation in eye banks—have shifted corneal surgery toward a more predictable, outcomes-driven discipline.
Yet Dr. Sawant noted that some of the most significant innovations aren’t always measured by volume. They emerge where clinical need is most urgent—particularly in treating limbal stem cell deficiency (LSCD), a complex, often debilitating condition that disproportionately affects patients in low- and middle-income regions.
KLAL-Pro: A Breakthrough in Treating Limbal Stem Cell Deficiency
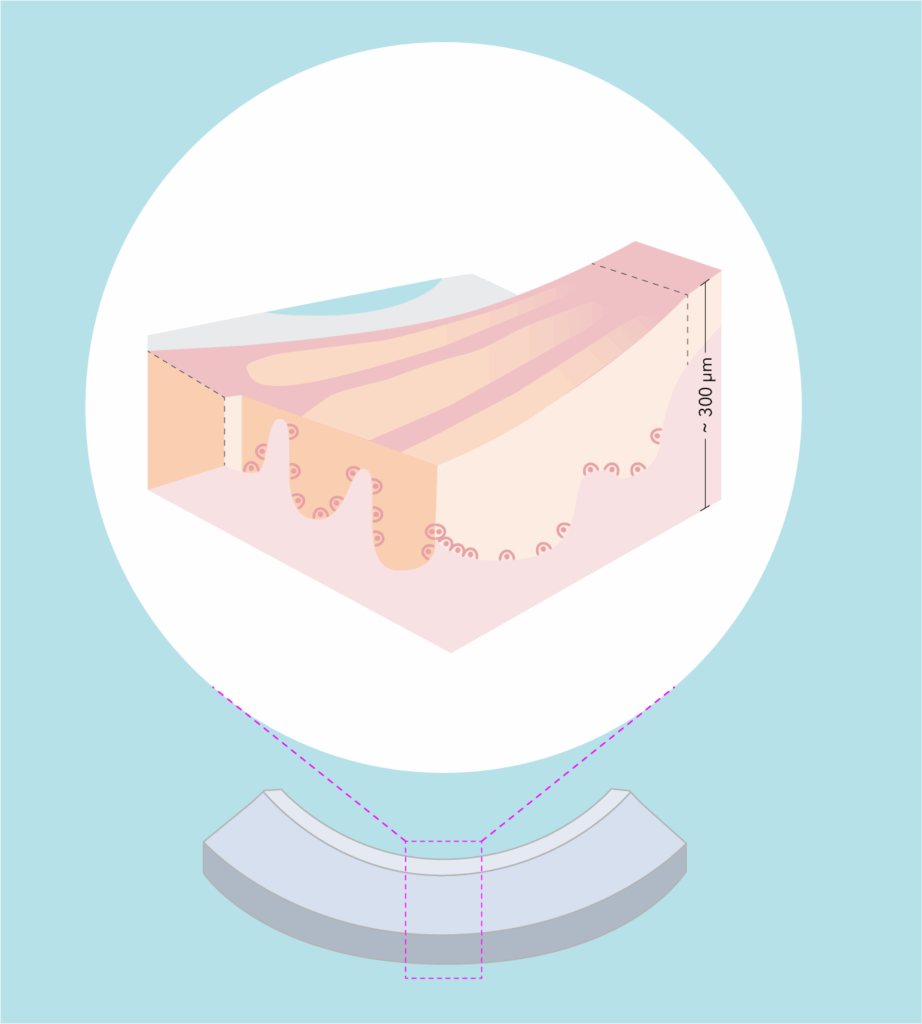
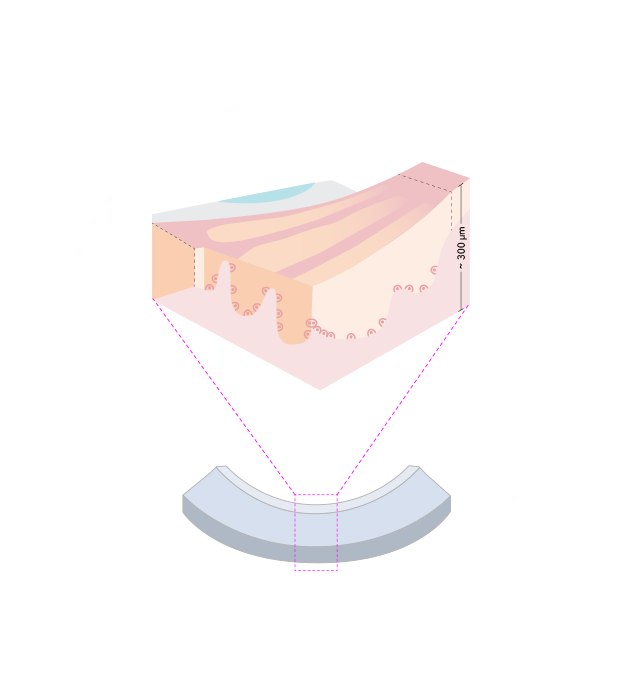
One of the most significant topics in our discussion was Eversight’s development of KLAL-Pro, an eye-bank–prepared, partial-thickness keratolimbal allograft enriched with limbal epithelial stem cells. As of April 2025, Eversight reports being the only eye bank preparing KLAL-Pro tissue, a ready-to-use keratolimbal allograft designed to reduce variability in tissue preparation and significantly reduce surgery time. 2
KLAL-Pro is:
- Customizable (up to 180° of donor limbus, 200–300 μm thickness) 3
- Prepared using a controlled, OCT-verified protocol 2
- Designed to preserve Palisades of Vogt-including limbal niche 2
- Optimized to support both unilateral and bilateral LSCD cases 3
- Can be efficiently utilized for Cincinnati and modified Cincinnati procedures 5 6
Early clinical experience suggests:
- Robust limbal stem cell expansion and re-epithelialization 2
- Significant reduction in surgery time compared to traditional KLAL alone 2
- Durability and regenerative potential that support long-term stability 2
Surgeons echo its value. As KLAL-Pro co-creator Dr. Nambi Nallasamy shared:
“KLAL-Pro grafts minimize excess stromal tissue and streamline ocular surface stem cell transplantation. With appropriate immunosuppression, my patients have had excellent outcomes.” 3
Insights From Eversight’s Recent Webinar: Modernizing LSCD Care
During the November 2025 Eversight webinar on LSCD management, Drs. Holland and Nallasamy emphasized the urgent need for more widespread awareness and adoption of evidence-based surgical interventions. Many patients with total LSCD continue to receive penetrating keratoplasties—despite poor outcomes for PKP in the presence of complete limbal stem cell failure.
Key takeaways from the webinar include:
1. Accurate Diagnosis of LSCD Is Critical
LSCD presents with a spectrum of signs—neovascularization, conjunctivalization, persistent epithelial defects—and should be assessed with clinical exam, impression cytology, confocal microscopy, or AS-OCT.
2. Procedural Choice Must Match Disease Severity
- Unilateral disease: conjunctival-limbal autograft (CLAU) 4
- Bilateral disease: KLAL or a Cincinnati procedure, which combines living-related tissue with KLAL to provide 360° limbal coverage 5 6
3. Immunosuppression Is Essential, and More Manageable and Safer Than Many Believe
Using protocols modeled after renal transplantation, long-term systemic immunosuppression dramatically increases graft survival. Importantly, across hundreds of LSCD surgical cases, no deaths were reported 7, debunking a longstanding misconception about the safety of systemic therapy.
4. KLAL-Pro Reduces OR Time and Complexity
A major barrier to LSCD surgery has been the technical burden of dissecting KLAL tissue. Eye-bank preparation shifts this workload upstream, enabling more surgeons to perform LSCD reconstruction confidently.
These insights provide important context for understanding where KLAL-Pro and other innovations fit within modern LSCD care.
Beyond Tissue: Where Innovation Is Heading Next
Our conversation with Dr. Sawant highlighted several emerging areas that promise to reshape corneal transplantation:
Cell-Based Therapies
Cultured endothelial cells combined with Rho Kinase (ROCK) inhibitors may one day reduce dependence on donor tissue altogether. Early results out of Japan and U.S. trials show strong promise.
AI and Advanced Imaging
Predictive tools for graft survival and donor selection, along with intraoperative OCT, are reducing variability and improving precision.
Improved Storage Media
Newer solutions—such as extended-life media validated in Eversight studies—may increase logistics flexibility and global tissue access.
Tissue Engineering & Bioscaffolds
Synthetic or decellularized matrices may eventually support corneal restoration where donor tissue is scarce.
You can read more from Dr. Sawant on these innovations and more in his recent perspective, Beyond tissue: How research & development are enhancing corneal transplantation.
A Shared Mission: Expanding the Surgical Toolkit
Perhaps the most resonant perspective came from Dr. Onkar Sawant, who articulated the field’s collective opportunity:
“Our goal should be to provide more tools in the toolkit for surgeons and eye doctors across the globe. Any innovation shouldn’t be seen as a threat to eye banking—but as an opportunity to broaden our impact in curing blindness.”
This philosophy aligns with Vision Share’s focus on collaboration, global access, and giving surgeons sight restoration options tailored to their patients’ needs.
Innovation Thrives When We Work Together
Our conversation with Eversight—and the insights shared through the Eversight Academy clinical education programs—highlight a consistent theme: the future of corneal transplantation depends on collaboration.
Eye banks, surgeons, researchers, and technology developers each play a vital role in strengthening access, safety, and outcomes. Whether through advances like DMEK, breakthroughs like KLAL-Pro, or emerging cell-based therapies, innovation continues to push the boundaries of sight restoration. Vision Share is proud to help bring these perspectives together and share them with the global community.
References
1. American Academy of Ophthalmology Cornea/External Disease Panel. Corneal edema and opacification preferred practice pattern®. American Academy of Ophthalmology. 2023.
https://www.aao.org/education/preferred-practice-pattern/corneal-edema-opacification-ppp-2023
2. Eversight. KLAL-Pro: Tissue Offerings & Services. Eversight; accessed Dec 2025. https://eversightvision.org/surgeons/tissue-offerings-services/klal-pro
3. Eversight. KLAL-Pro: A Breakthrough in Treating Limbal Stem Cell Deficiency (LSCD). Eversight Blog; accessed Dec 2025.
https://eversightvision.org/blog/klal-pro-a-breakthrough-in-treating-limbal-stem-cell-deficiency-lscd
4. Eversight Academy. A Complete Approach to Limbal Stem Cell Deficiency: KLAL-Pro and Postoperative Management Strategies. YouTube; published 2025. https://www.youtube.com/watch?v=FHBJpstGkWg&t=4s
5. Biber, Joseph M MD; Skeens, Heather M MD; Neff, Kristiana D MD; Holland, Edward J MD. The Cincinnati Procedure: Technique and Outcomes of Combined Living-Related Conjunctival Limbal Allografts and Keratolimbal Allografts in Severe Ocular Surface Failure. Cornea 30(7):p 765-771, July 2011. | DOI: 10.1097/ICO.0b013e318201467c
6. Chan, Clara C. MD, FRCSC*,†; Biber, Joseph M. MD‡; Holland, Edward J. MD*,†. The Modified Cincinnati Procedure: Combined Conjunctival Limbal Autografts and Keratolimbal Allografts for Severe Unilateral Ocular Surface Failure. Cornea 31(11):p 1264-1272, November 2012. | DOI: 10.1097/ICO.0b013e31823f8e95
7. Holland EJ, Mogilishetty G, Skeens HM, Hair DB, Neff KD, Biber JM, Chan CC.
Systemic immunosuppression in ocular surface stem cell transplantation: results of a 10-year experience. Ophthalmology. 2012;119(4):651–658. doi:10.1016/j.ophtha.2011.09.045. PMID: 22333664.
About Vision Share
Vision Share is a 501(c)(3) nonprofit organization and proudly represents the world’s largest network of eye banks. Each member eye bank is Eye Bank Association of America (EBAA) certified and registered with the Food and Drug Administration (FDA). Guided by a shared vision, Vision Share pools resources to provide surgeons with dependable, high-quality ocular tissue tailored to their specific needs. The Vision Share network has delivered over 141,000 corneas worldwide, transforming lives and restoring sight.
